Gas Chromatography includes all those chromatographic processes in which a solid or a liquid coated on a solid support is used as the stationary phase and gas is used as the mobile phase. In the Gas chromatographic technique, the components of the mixture to be separated are dissolved in a suitable organic solvent and placed on top of the column of the stationary phase by an injection device. They are then carried or transported through the stationary phase in the gaseous or vapor form. Thus this separation technique can be applied to separating only those substances, which can be vaporized at the operating temperatures.
Classification
Based on the principle of separation and the type of the stationary phase the Gas chromatographic techniques can be broadly divided into two as follows:
1. Gas-Solid Chromatography (GSC) or Adsorption Gas Chromatography, where a solid (usually an adsorbent) is used as the stationary phase and a neutral gas as the mobile phase.
2. Gas-Liquid Chromatography (GLC) or Partition Gas chromatography, where a liquid, coated on an inert support, is used as the stationary phase and an inert gas as the mobile phase.
Principle and Technique
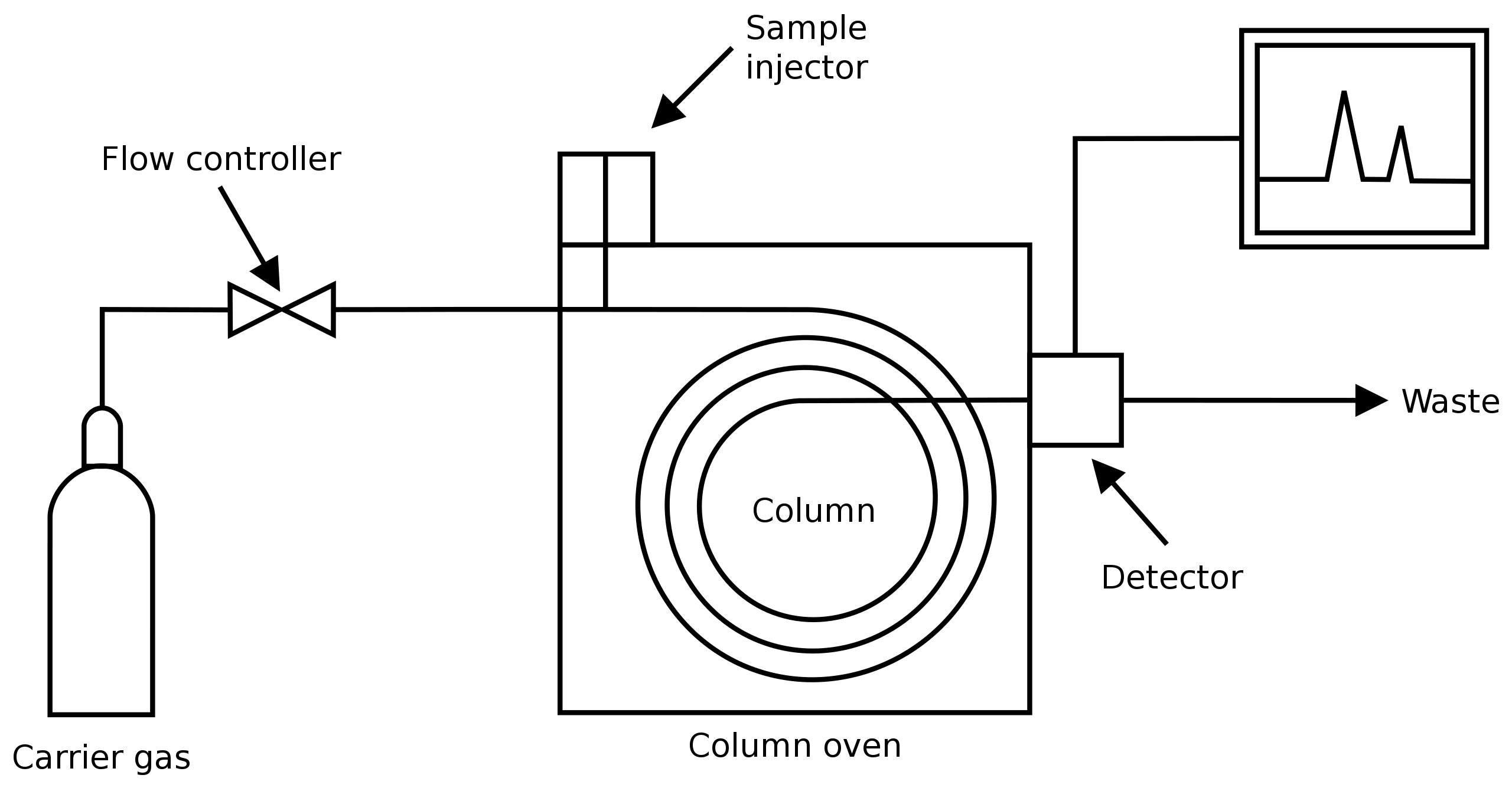
The stationary phase in gas chromatography is used as a column, which is packed in a glass or metallic tube. Its internal diameter ranges from 2 to 10 mm and the length from I to 20 m. The liquid, solid, or gaseous substance to be separated is placed on one end of the column by means of a suitable injection device and then driven through the column by an inert carrier gas (the mobile phase). The rate of flow of the mobile phase is adjusted to a constant speed that may range from 0.3 to 10 liter per hour. The components of the mixture are separated as they pass through the column, and the separated fractions gradually emerge at the other end of the column along with the mobile phase. The substances in the emerging mobile phase are then detected or characterized by measuring a physical or chemical property in a detector device, usually a fathometer. This measurement is continuously recorded on a moving chart by a recording device. There thus results a diagram in which the observed figures are plotted against time. This plotted chart or diagram is the gas chromatogram.
The separated components are identified in terms of their Retention time (Rt). Retention time of a compound is the interval of time between the introduction of the mixture into the column and the elution of that compound from the column.
Gas Chromatography Assembly
The basic components of a Gas Chromatographic assembly include a gas cylinder or a gas generator with a control valve, an injection device, a thermostat oven in which is accommodated the column, a detector device directly connected to the column and an amplifier and recorder connected to the detector.
Application of Gas Chromatography
Gas chromatography, particularly the GLC, has been very successfully used both as a qualitative and quantitative method for analysis of a large number of natural substances of pharmaceutical importance, such as volatile oils, camphor, plant acids, some alkaloids, resins, sapogenins and cardioactive glycosides and aglycones.

